VedaNadhi
Today's Question
Living World - Biodiversity
(1) Linnaeus
(2) Darwin
(3) Lamarck
(4) Huxley
(4) Huxley
Sir Julian Huxley introduced the term 'New Systematics'. New Systematics aims at taking into consideration, all characteristics of organisms gathered from the study of ecology, genetics, cytology, physiology, biochemistry, behaviour, etc., for systematics.
Chemical Coordination
(1) Cholesterol
(2) 7-hydroxy cholesterol
(3) Cephalin cholesterol
(4) All of the above
vitamin D is a steroid, which is synthesized from cholesterol. Vitamin-D2 is formed from plant sterol, ergo sterol, which occurs in yeast and other fungi in the presence of ultraviolet light. Vitamin-D3 formed from animal sterol, 7-dehydroxy cholesterol, which occurs in animal skin. Its synthesis also needs ultraviolet light
Plant Kingdom
(1) Algae can be classified according to their pigments
(2) All algae are filamentous
(3) Spirogyra does not produce zoospores
(4) Most algae are photosynthetic
(2) All algae are filamentous
Algae include organisms that are unicellular, colonial, filamentous, or large and multicellular.
Animal Kingdom
(1) Flagellata
(2) Sporozoa
(3) Rhizopoda
(4) Ciliata
(2) Sporozoa
Members of class Sporozoa have no specialised locomotory organelles and contractile vacuoles.
Morphology of Flowering Plants
(1) Monocarpic
(2) Bicarpic
(3) Polycarpic
(4) Apocarpic
(1) Monocarpic
Monocarpic refers to a plant that flowers only once during its life, after fruiting, the leaves senescence and the plant dies completely.
Anatomy Flowering plants
(1) Gymnosperms
(2) Dicotyledons
(3) Pteridophytes
(4) Monocotyledons
(1) Gymnosperms
A pit possessing an extension of the secondary wall, i.e. a border, arching over part of the pit cavity is called bordered pit. Bordedred pits mainly occur in vessel elements, tracheids and fibres in gymnosperm xylem.
Structural Organisation in Animals
(1) Human intestine
(2) Mammalian liver
(3) Graffian follicles
(4) Pancreatic acini
(1) Human intestine
Lamina propria is a layer of loose connective tissue in the mucosa of gut wall, which houses the bases of glands and contains blood and lymph vessels.
Cockroach
(1) Sternum
(2) Muscles
(3) Pleuron
(4) Mesenteries
(3) Pleuron
Pleuron is the name given to the lateral part of the cuticle in each segment of a cockroach.
Structure of a Cell
(1) DNA and histones
(2) DNA but no histones
(3) Neither DNA nor histones
(4) Either DNA or histones
(2) DNA but no histones
Histones are not found in association with the DNA of prokaryotes.
Biomolecules
(1) Mineral salts absorbed by the plant
(2) Organic matter of the plant
(3) Both the mineral salts and organic matter
(4) None of the above
(1) Mineral salts absorbed by the plant
Plant ash is an indication of mineral salts absorbed by the plant. On heating dry plant or a plant part at 600$^o$ C in a crucible, the organic matter dissociates. The ash, which is left, contains oxides and carbonates of elements. About 60 elements have been reported from plant ashes. Out of these, 30 are present in all plants. Again out of these 30, 16 are essential, rest are nonessential.
Cell cycle
(1) Cleavage of the nucleus without recognisable chromosomes
(2) A division of chromosomes
(3) An indefinite division of chromosomes
(4) Chromosomes are present at the bridge
(1) Cleavage of the nucleus without recognisable chromosomes
Amitosis is an unusual process of nuclear division in which the nucleus is pinched into two by a constriction. The details of how it occurs are uncertain. Amitosis occurs in some protozoans ( including the macromolecules of the Ciliata) and in the embryonic membranes.
Transport in Plants
(1) Respiratory activity of the root
(2) Tension on cell sap due to transpiration
(3) Tissue organisation
(4) None of the above
(1) Respiratory activity of the root
Active absorption of water from the soil by the roots involves an expenditure of metabolic energy, which comes from the respiring cells of the root.
Mineral Nutrition in plants
(1) Is present in plant ash
(2) Is available in soil
(3) Is indispensable for growth
(4) Improves plants growth
(3) Is indispensable for growth
An essential element is one, which is indispensable for growth. D.I. Arnon and P.R. Stout gave criteria of essentiality of an element in 1939.
Photosynthesis in Higher Plants
(1) $C_6H_{12}O_6$ containing $^{18}$O
(2) $H_2O$ containing $^{18}$O
(3) $CO_2$ containing $^{18}$O
(4) Oxygen in the form of ozone
(2) $H_2O$ containing $^{18}$O
In 1940 Sam Ruben used an isotope of oxygen, $^{18}$O to find out where the oxygen atoms went in photosynthesis. Ruben fed plants water-containing $^{18}$O but because $^{18}$O is not a radioactive isotope of the most common form of oxygen, $^{16}$O. Ruben used a mass spectrometer to determine the fate of the $^{18}$O. The $^{18}$O was found in the oxygen gas produced by the plant and none in the glucose formed.
Respiration in Higher Plants
(1) Energy is stored in the form of ADP
(2) Energy is released and stored in the form of ATP
(3) Energy is not released at all
(4) Energy is used up
(2) Energy is released and stored in the form of ATP
Cellular respiration is energy releasing enzymatically controlled catabolic process, which involves a step-wise oxidative breakdown of organic substances inside living cells.
Plant Growth & Development
(1) S
(2) Mn
(3) Zn
(4) K
(3) Zn
Zinc is essential for the synthesis of tryptophan-a precursor of auxins.
Digestion and Absorption
(1) Dehydration synthesis
(2) Hydrolysis
(3) Absorption
(4) Assimilation
(2) Hydrolysis
Digestion is the process of breaking down of the covalent bonds in the organic food molecules (Carbohydrates, lipids, proteins and nucleic acids) by hydrolysis. It is catalysed by digestive enzymes. The simpler and smaller molecules thus produced can then get entry into the surrounding cells.
Breathing & Exchange of Gases
(1) Tracheae
(2) Malphigian tubules
(3) Gills
(4) Book lungs
(3) Gills
Typically gills are respiratory structures in aquatic animals (both vertebrates and invertebrates). Gills possess a large surface area for extensive gas exchange.
Body Fluids and circulation
(1) Cytoplasm
(2) Interstitial fluid
(3) Digestive cavity
(4) Urine
(2) Interstitial fluid
When the blood does not remain confined to the blood vessels and flows into spaces and channels, called lacunae and sinuses in the tissues, it is termed as the open circulatory system. The open circulatory system is common to molluscs and arthropods.
Excretory products and their elimination
(1) Gut
(2) Coelom
(3) Lymph
(4) Blood
(2) Coelom
In Annelids like Nereis, earthworm, leech, etc., the tubular coiled structures, the nephridia are excretory organs, which drains materials directly from the coelom.
Locomotion and Movement
(1) 126 bones
(2) 100 bones
(3) 103 bones
(4) 80 bones
(4) 80 bones
The axial skeleton consists of the skull, the spine, the ribs and the sternum (breastbone) and includes 80 bones.
Neural control and Coordination
(1) Nucleus
(2) Sarcolemma
(3) Axon
(4) Cytoplasm
(2) Sarcolemma
The sarcolemma is the name of the cell membrane of a muscle fibre or muscle cell.
Chemical Coordination
(1) Prostaglandin
(2) Estrogen
(3) Epinephrine
(4) Progesterone
(3) Epinephrine
The hormones epinephrine and norephinephrine secreted by the adrenal medulla and thyroxine by thyroid gland are derived from the amino acid tyrosine.
Reproduction in Organisms
(1) Citrus
(2) Apples
(3) Rubber
(4) All of these
(4) All of these
Grafting is routinely used for the propagation of rubber, apple, citrus,pear, mango and guava.
Sexula Reproduction in Flowering Plants
(1) Layering
(2) Cutting
(3) Grafting
(4) Micropropagation
(3) Grafting
Scion is a portion of a plant, usually a piece of young stem, which is inserted into a rooted stock in grafting.
Human Reproduction
(1) Whale
(2) Bat
(3) Penguin
(4) Mouse
(3) Penguin
Oviparity is the production of undeveloped eggs, which are laid or spawned by the female.
Principles of Inheritance and Variation
(1) Austria
(2) Germany
(3) Britain
(4) Sweden
(1) Austria
Gregor Mendel was born in Heinzendorf, Austria on July 22,1822.
Molecular basis of Inheritance
(1) Transformation of rough coated strain of Diplococcus pneumoniae into smooth coated strain
(2) The establishmmennt of DNA as the chief chemical constituent of chromosomes
(3) The establishment of DNA is a self-replicating substance
(4) Transduction of bacteria by action of bacteriophage
(2) The establishmmennt of DNA as the chief chemical constituent of chromosomes
Transformation is a permanent genetic change induced in cell following incorporation of new DNA.
Evolution
(1) Organisms arose from other organism
(2) Organism arose from air
(3) Organism arose abiogenetically
(4) Organism arose simultaneously
(3) Organism arose abiogenetically
According to Theories of spontaneous generation, life has originated from non-living matter abiogenetically, i.e. without the intervention of living things.
Human Health and Diseases
(1) Hay fever
(2) Enteric fever
(3) Skin cancer
(4) Goitre
(1) Hay fever
Hay fever. Inflammation in the nasal passage and eyes caused by an allergic reaction to flowers and their pollen and scent, also to dust.
Strategies for Enhancement in Food Production
(1) Selection
(2) Inbreeding
(3) Interspecific hybridisation
(4) Cross-breeding
(3) Interspecific hybridisation
Mule refers to hybrid offspring of a male donkey and a female horse. Mules are usually sterile.
Microbes in Human Welfare
(1) Aureomycin
(2) Neomycin
(3) Terramycin
(4) Erythromycin
(2) Neomycin
Neomycin was first isolated in 1949 by the American microbiologist Selman Waksman from a strain of the bacterial species Streptomyces fradiae
Biotechnology: principles and Processes
(1) Phage vector
(2) Bacterial plasmid
(3) Yeast plasmid
(4) Cosmids
(4) Cosmids
Cosmids are DNA vectors that allow the insertion of long fragments of DNA (up to 50 kbases).
Biotechnology and its Application
(1) They are found in viruses
(2) They are main parts of chromosomes
(3) They are widely used in gene transfer
(4) They contain genes for vital activities
(3) They are widely used in gene transfer
Plasmids are extra chromosomal circular DNA replicates independently. Plasmids that can get associated temporarily with nucleoid are known as episomes.
Organisms and Populations
(1) Vegetation
(2) Community
(3) Flora
(4) Population
(3) Flora
All the plant life normally present in a given habitat at a given time is called flora.
Ecosystem
(1) Erect
(2) Inverted
(3) Both (1) and (2)
(4) None of the above
(1) Erect
Pyramid of numbers show relationship between the numbers of primary producers and consumers of different orders. Examples: Upright pyramids of numbers-grassland ecosystem; cropland ecosystem. Inverted pyramid of numbers-Tree.
Biodiversity and Conservation
(1) Madagascar
(2) Mauritius
(3) Sri Lanka
(4) India
(1) Madagascar
The endangered largest living lemur Indri indri is found in Madagascar
Environmental Issues
(1) $CO_2$, $O_2$, $NO_2$, $NH_3$
(2) Chlorofluorocarbons, $CO_2$, $NH_3$, $N_2$
(3) $CH_4$, $N_2$, $CO_2$, $NH_3$
(4) Chlorofluorocarbons, $CO_2$, $CH_4$, $NO_2$
(4) Chlorofluorocarbons, $CO_2$, $CH_4$, $NO_2$
Greenhouse gases. Atmopheric gases such as carbon dioxide, methane, chlorofluorcarbons,nitrous oxides, ozone and water vapour that slow the passage of re-radiated heat through the Earth's atmosphere by absorbing infrared radiation.
Living World
(1) It is based on evolutionary relationship of plants
(2) It is based on similarities and differences in floral and other morphological characters only
(3) It took into account the physiological facts along with the morphological characters
(4) None of the above
(2) It is based on similarities and differences in floral and other morphological characters only
Linnaeus was the first to recognise the significance of flower and fruit structure. This system of classification is mainly based on the number, size and union of stamens and carpels, so this system is often called sexual system of classification.
Biological Classification
(1) Safranin and iodine
(2) Crystal violet and cotton blue
(3) Cotton blue and iodine
(4) Crystal violet and iodine
(4) Crystal violet and iodine
Differential staining procedures divide bacteria into separate groups based on staining properties.
The Gram stain, developed in 1884 by the Danish physician Christian Gram, is the most widely employed staining method in bacteriology.
It is a differential staining procedure because it divides bacteria into two classes: Gram-negative and Gram-positive.
In the first step of the Gram staining procedure, the smear is stained with the basic dye crystal violet.
It is followed by the treatment with an iodine solution functioning as a mordant. That is, the iodine increases the interaction between the cell and the dye so that the cell is stained strongly.
The smear is next stained decolourised by washing with alcohol or acetone.
Gram-positive bacteria retain the crystal violet, whereas Gram-negative bacteria lose their crystal violet and become colourless.
Finally, the smear is counterstained with safranin. Safranin colours Gram-negative bacteria pink to red and leave Gram-positive bacteria dark purple.
Plant Kingdom
(1) Homogamy
(2) Hologamy
(3) Syngamy
(4) Autogamy
(2) Hologamy
Condition of having gametes similar to somatic cells or fusion between mature individuals of a species is called hologamy.
Animal Kingdom
(1) Has abundance of proteins
(2) Constantly changes its body shape
(3) Does not change its body shape
(4) Has protoplasm
(2) Constantly changes its body shape
The name Amoeba proteus has been derived from two Greek words Amoeba from amoibe, which means change and proteus after the name of the mythical sea-god Proteus, who could change shape.
Morphology of Flowering Plants
(1) Santalum
(2) Loranthus
(3) Viscum
(4) Cuscuta
(1) Santalum
Sandalwood tree (Santalum album) is an example of partial root parasite found abundantly in karnataka.
Anatomy of Flowering Plants
(1) Terminal
(2) Intercalary
(3) Absent
(4) Sub-terminal
(4) Sub-terminal
The meristem in the root is subterminal because it is covered by the root cap.
Structural Organisation in Animals
(1) Squamous epithelium
(2) Columnar epithelium
(3) Glandular epithelium
(4) Ciliated epithelium
(1) Squamous epithelium
The peritonium is thin membrane that lines the abdominal and pelvic cavities and covers most abdominal viscera. It is composed of layer of mesothelium supported by a thin layer of connective tissue.
Cockroach
(1) Mesenteron
(2) Crop
(3) Gizzard
(4) Ileum
(1) Mesenteron
The internal lining of the wall of midgut or mesenteron in cockroach is an endodermal epithelium of columnar cells. The epithelium is internally covered, not by a cuticle, but by a thin transparent peritrophic membrane formed of chitin and protein.
Structure of a Cell
(1) Proper nucleus
(2) Endoplasmic reticulum & Mitochondria
(3) Proper nucleus and most of cell organelles
(4) Cell Wall
(3) Proper nucleus and most of cell organelles
Prokaryote is an organism in which the genetic material is not enclosed in a cell nucleus. Bacteria are prokaryotes.
Biomolecules
(1) Chlorine
(2) Zinc
(3) Manganese
(4) Iron
(4) Iron
Iron is constituent of ferredoxin and cytochromes; required for synthesis of chlorophyll.
Cell Cycle
(1) M-phase
(2) S-phase
(3) G$_1$ phase
(4) G$_2$ phase
(2) S-phase
Histone is a protein associated with nucleic acid in chromatin of eukaryotic cells. Histones are characterised by high levels of the basic amino acids arginine and lysine. Histones are not found in association with the DNA of prokaryotes. The bulk of the histones are synthesised during S phase of the cell cycle.
Transport in Plants
(1) 40-45$^o$ C
(2) 20-35$^o$ C
(3) 5-10$^o$ C
(4) 10-15$^o$ C
(2) 20-35$^o$ C
The optimum temperature for water absorption is between 20$^o$ C to 35$^o$ C
Mineral Nutrition in Plants
(1) Carbon
(2) Nitrogen
(3) Phosphorous
(4) Hydrogen
(1) Carbon
Plants obtain carbon from carbon dioxide in the atmosphere, oxygen and hydrogen primarily from the water they take in through their roots; nitrogen from mainly biologically fixed ions present in the soil and their other nutrients from the soil.
Photosynthesis in Higehr plants
(1) Crops
(2) Phytoplanktons of the sea
(3) Forests
(4) Great land area
(2) Phytoplanktons of the sea
Algae of oceans carry out nine-tenth of all photosynthesis of the world.
Respiration in Plants
(1) 34 molecules of ADP into 34 molecules of ATP
(2) 30 molecules of ADP into 30 molecules of ADP
(3) 38 molecules of ADP into 38 molecules of ATP
(4) 32 molecules of ADP into 32 molecules of ADP
(3) 38 molecules of ADP into 38 molecules of ATP
Oxidation of one molecule of glucose in aerobic respiration results in the formation of 38 ATP molecules. In most eukaryotic cells 2 ATP molecules are consumed in transporting 2 NADH produced during glucolysis, into the mitochondria for further oxidation. Thus, in such cells, there is net gain of 36 ATP instead of 38 ATP molecules.
Plant Growth and Development
(1) Auxin increases
(2) Auxin decreases
(3) Abscisic acid decreases
(4) Gibberellic acid decreases
(2) Auxin decreases
Auxin is a controlling factor in abscission. Reduction in the auxin content in leaves and fruits results in their abscission. Application of auxin like 2,4-D, IAA, NAA to the leaves and fruits prevent their premature falling
Digestion and Absorption
(1) Membranes
(2) Cell Wall
(3) Connective tissues
(4) Muscle bundle
(2) Cell Wall
Chewing is especially important for most fruits and raw vegetables, because these have indigestible cellulose wall around their nutrient portions, which must be broken, before enzymes can act upon the food.
Respiration in Animals
(1) Plasma
(2) Erythrocytes
(3) Leucocytes
(4) Neurons
(2) Erythrocytes
Carbonic anhydrase is an enzyme, present in red blood cells. It catalyses the reaction between carbon dioxide and water to form carbonic acid, which subsequently dissociates. This reaction is one of the fastest known and controls the elimination of carbon dioxide from the body and the pH of urine. It also facilitates the transfer of carbon dioxide from the tissues to the blood and from the blood to the alveoli (air sacs) of the lungs.
Body Fluids and Circulation
(1) Fishes
(2) Reptile
(3) Birds
(4) Amphibians
(1) Fishes
In fishes, heart is two chambered with an auricle and ventricle; it receives venous blood only and pumps blood to gills only for oxygenation. Hence, referred to as the venous heart or branchial heart.
Excretory Products and their Elimination
(1) Citrulline and Urea
(2) Ornithine and Ammonia
(3) Ornithine and Urea
(4) Citrulline and Ammonia
(3) Ornithine and Urea
The enzyme arginase catalyses the hydrolysis of arginine to ornithine and urea and is important in the urea cycle.
Locomotion and Movement in Animals
(1) 10 bones
(2) 22 bones
(3) 30 bones
(4) 24 bones
(2) 22 bones
The skull of man is made up of 22 bones of varying shapes and sizes. The cranium consists of eight flat plate-like bones that surround and protect the brain. Some of these contribute to the formation of the facial skeleton. The remaining 14 bones, including the mandible, form the face. The mandible is the only movable bone in the skull; the rest are connected by immovable joints called sutures.
Neural Control and Coordination
(1) Satellite cells
(2) Neuroglia
(3) Schwann cells
(4) All of the above
(2) Neuroglia
Neuroglia are supporting cells, which form packing around the neurons. There are present three types of neuroglia: astrocytes, oligodendrocytes and microgliocytes.
Chemical Control and Coordination
(1) Insulin
(2) Tyrosine
(3) Glycogen
(4) Cholesterol
(4) Cholesterol
The hormones secreted by the adrenal cortex, testes, ovaries and placenta are composed of steroids.
Reproduction in Organisms
(1) From root cuttings
(2) Without fusion of gametes
(3) From fusion of gametes
(4) From stem cutting
(2) Without fusion of gametes
Apomixis is a form of asexual reproduction, including vegetative propagation. The term is very often used in narrower sense to mean the production of seeds without fertilisation.
In this restricted sense the term is synonymous with agamospermy.
An apomictic plant is an organism, which chiefly reproduces this way. Apomixis is a natural way of cloning plants through seed. It offers plant breeders a unique system for developing new and distinctive cultivars in many species. Apomixis is widespread among several plant families including Gramineae, Compositae and Rosaceae.
Sexual Reproduction in Flowering Plants
(1) Sugarcane
(2) Coffee
(3) Citrus
(4) All of these
(4) All of these
Cuttings are small pieces of stem, root or leaves which when placed in the soil can sprout to form new plants. Stem cuttings are commonly used in sugarcane, coffee, citrus, tea, Duranta, Rosa and Clerodendron.
Human Reproduction
(1) 200
(2) 2000
(3) 200,000
(4) 200,000,000
(4) 200,000,000
The normal human male usually produces 200,000,000 sperms per ejaculation, assuring an adequate number for fertilisation to take place. Fewer than 100 ever reach the vicinity of the egg. However only one sperm normally enters an egg.
Reproductive Health
(1) Fertilisation is done outside body
(2) Foetus is grown in a test tube
(3) Fertilisation is done inside body
(4) None of these
(1) Fertilisation is done outside body
Patient's eggs and her partner's sperm are collected and mixed together in a laboratory to achieve fertilisation outside the body. The embryos produced may then be transferred into the female patient.
Principles of Inheritance and Variation
(1) Epistatics
(2) Inhibition
(3) Quantitative Inheritance
(4) Incomplete Dominance
(4) Incomplete Dominance
Incomplete dominance in Mirabilis jalapa (four-O' clock plant) was reported by Carl Correns in 1905.
Molecular Basis of Inheritance
(1) J.D. Watson
(2) Hershey and Chase
(3) Alfred Griffith
(4) Boveri and Sutton
(2) Hershey and Chase
Alfred Day Hershey was awarded the Nobel Prize in 1969 for the famous "blender's experiment".
Hershey and his assistant Martha Chase showed that only DNA and not protein, was injected into a bacterial cell by an infecting phage particle. The DNA was sufficient to transfer to the bacteria all the genetic information needed to produce more phage.
Evolution
(1) Laboratory preserved animal
(2) Dead animal of the past
(3) Inorganic relic of the past
(4) Stuffed animal
(2) Dead animal of the past
Fossil is the remains or traces of any organism that lived in the geological past. In general only the hard parts of organisms become fossilized (e.g., bones, teeth, shells and wood) but under certain circumstances the entire organism is preserved.
Human Health and Disease
(1) Interferons
(2) Cytotoxins
(3) Antibiotics
(4) Anticancer protein
(1) Interferons
Interferons are a group of proteins called cytokines produced by white blood cells, fibroblasts, or T-cells as part of an immune response to a viral infection or other immune trigger. The name of the proteins comes from their ability to interfere with the production of new viral particles.
Strategies for Enhancement in Food Production
(1) Protoxins
(2) Phytoalexins
(3) Prolactins
(4) All the above
(2) Phytoalexins
Phytoalexins are low molecular weight antimicrobial compounds produced by plants after infection or metabolic stress. They are part of a plant's multicomponent response mechanism for disease resistance and the speed and magnitude of their accumulation is important in determining disease resistance to fungal and bacterial diseases.
Microbes in Human Welfare
(1) Bacillus megaterium and Xanthomonas sp.
(2) Bacillus subtilis and E.coli
(3) Rhizobium meliloti and Azotobacter
(4) Lactobacillus casei and Streptococcus thermophilus
(4) Lactobacillus casei and Streptococcus thermophilus
Yoghurt is a fermented dairy product made by adding bacterial cultures to milk, which causes the transformation of the milk's sugar, lactose into lactic acid. This process gives yoghurt its refreshingly tart flavour and unique pudding-like texture.
Biotechnology: Principles and processes
(1) Agrobacterium tumefaciens
(2) Protoplasm fusion
(3) Transposons
(4) Microinjection
(1) Agrobacterium tumefaciens
Plant transformation mediated by Agrobacterium tumefaciens, a soil plant pathogenic bacterium, has become the most used method for the introduction of foreign genes into plant cells and the subsequent regeneration of transgenic plants.
Biotechnology and its Applications
(1) Traditional knowledge
(2) Biomolecules and regarding bioresources, genes isolated from bioresources
(3) Bioresources
(4) All the above
(4) All the above
Biopiracy refers to the exploitation, through use of biopatents of bioloical resources or bioresources of other nations without proper authorisation from the countries concerned.
Organisms and Populations
(1) The Environment
(2) The Biosphere
(3) The Natural Ecosystem
(4) A Community
(2) The Biosphere
Noosphere is referred to a human-dominated environment. It is another name that has been given to the biosphere since man has changed it from a naturally self-sufficient system to a system of resources for himself.
Ecosystem
(1) Synecology
(2) Autecology
(3) Habitat ecology
(4) Population ecology
(2) Autecology
Autecology is the study of an individual species in its environment.
Biodiversity and Conservation
(1) Biota of Red Sea
(2) Effect of red light on photosynthesis
(3) Red pigmented plants
(4) Threatened species
(4) Threatened species
Red Data Book: A catalogue published by the International Union for the Conservation of Nature and Natural Resources (IUCN) that lists rare species and those in danger of extinction.
Environmental Issues
(1) Petiole but not in lamina
(2) Leaf tip and leaf margins
(3) Stem tips only
(4) Mid rib and Lamina
(2) Leaf tip and leaf margins
Exposure of plants to high fluoride concentration results in chlorosis and necrosis at the tips and margins of leaves.
Digestion and Absorption
(1) A mass of crushed food moistened with saliva
(2) The semisolid material resulting from partial digestion in the stomach
(3) The milky emulsified fat absorbed from small intestine
(4) Indigestible materials that helps in movement and absorption
(1) A mass of crushed food moistened with saliva
A bolus is a ball of chewed food bound together with saliva that is formed in the mouth by the action of the tongue. The bolus is shaped to a size that allows it to pass into the oesophagus after being swallowed.
Breathing and Exchange of gases
(1) Pharynx
(2) Glottis
(3) Bronchial tube
(4) Larynx
(4) Larynx
Larynx is an organ, situated at the front of the neck above the windpipe, which contains the vocal cords, responsible for the production of vocal sounds.
Body Fluids and Circulation
(1) Parietal cells
(2) Haemocytes
(3) Oxyntic cells
(4) Endothelial cells
(4) Endothelial cells
The cells constituting walls of the blood capillaries are known as endothelial cells.
Excretory Products and their Elimination
(1) Pelvis
(2) Ileum
(3) Seminiferous tubules
(4) Cystic duct
(1) Pelvis
The human kidney measures about 10 cm in length, about 5 cm in breadth and about 9 cm in thickness. On the concave side of the kidney, there is a longitudinal opening called the hilum and to this area the blood vessels, nerves and ureter join kidneys at extensive, flat, funnel shaped space called the renal pelvis.
Locomotion and movement
(1) Lateral side of skull
(2) Neural canal
(3) Anterior side of skull
(4) Posterior side of skull
(4) Posterior side of skull
Foramen magnum is the large opening at the hind-end of a vertebrate skull through which the spinal cord passes to the brain.
Neural control and coordination
(1) Brain
(2) Spinal cord
(3) Tongue
(4) Retina
(1) Brain
The adult human brain contains more than a thousand million neurons. It has been estimated that in the cerebral cortex alone.
Chemical coordination
(1) The posterior pituitary gland
(2) Lymphatic vessels
(3) Blood vessels
(4) Axons
(3) Blood vessels
Neurons in the hypothalamus secrete releasing hormones into veins that carry the releasing hormones directly to the vessels of the adenohypophysis. Thus by-passing the normal circulatory route
Some Basic Concepts of Chemistry
(1) 2.8 kg
(2) 6.4 kg
(3) 9.6 kg
(4) 96 kg
(3) 9.6 kg
$C_2H_4(28g) + 3O_2(96g) → 2CO_2 + 2H_2O $
For complete combustion,
2.8 kg of $C_2H_4$ requires = $\frac{96}{28} × 2.8 × 10^3$ g
= $9.6 × 10^3$ g = 9.6 kg of $O_2 $
Structure of Atom
(1) $H^+ $
(2) $Li^+ $
(3) $Na^+ $
(4) $Mg^{2+} $
(2) $Li^+ $
Species - No. of electrons
$Be^{2+}$ - 2
$H^+$ - 0
$Li^+$ - 2
$Na^+$ - 10
$ Mg^{2+}$ - 10
Classfification of Elements & Periodicity in Properties
(1) Carbon family, $[Rn] 5f^{14} 6d^{10} 7s^2 7p^2$
(2) Oxygen family, $[Rn] 5f^{14} 6d^{10} 7s^2 7p^4$
(3) Nitrogen family, $[Rn] 5f^{14} 6d^{10} 7s^2 7p^6$
(4) Halogen family, $[Rn] 5f^{14} 6d^{10} 7s^2 7p^5$
(1) Carbon family, $[Rn] 5f^{14} 6d^{10} 7s^2 7p^2$
The electronic configuration of the element with Z = 114 (Flerovium) is $[Rn] 5f^{14} 6d^{10} 7s^2 7p^2$.Hence, it belongs to carbon family which has the same outer electronic configuration.
Chemical Bonding & molecular Structure
(1) Ammonia, beryllium difluoride, water, 4-dichlorobenzene
(2) Boron trifluoride, hydrogen fluoride, carbon dioxide, 1, 3-dichlorobenzene
(3) Nitrogen trifluoride, beryllium difluoride, water, 1, 3-dichlorobenzene
(4) Boron trifluoride, beryllium difluoride, carbon dioxide, 1, 4-dichlorobenzene
(4) Boron trifluoride, beryllium difluoride, carbon dioxide, 1, 4-dichlorobenzene
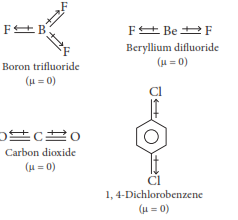
States of Matter
(1) Covalent $<$ hydrogen bonding $<$ van der Waals’ $<$ dipole-dipole
(2) van der Waals’ $<$ hydrogen bonding $<$ dipole-dipole $<$ covalent
(3) van der Waals’ $<$ dipole-dipole $<$ hydrogen bonding $<$ covalent
(4) Dipole-dipole $<$ van der Waals’ $<$ hydrogen bonding $<$ covalent.
(2) van der Waals’ $<$ hydrogen bonding $<$ dipole-dipole $<$ covalent
The strength of interaction follows the order : van der Waals’ $<$ hydrogen-bonding $<$ dipole-dipole $<$ covalent. It is so because bond length of H-bond is larger than that of a covalent bond.And also covalent bond is strongest because, the greater the extent of overlapping, the stronger is the bond formed.
Thermodynamics
(1) $∆$E = W ≠ 0, q = 0
(2) $∆$E = W = q ≠ 0
(3) $∆$E = 0, W = q ≠ 0
(4) W = 0, $∆$E = q ≠ 0
(1) $∆$E = W ≠ 0, q = 0
The mathematical form of first law of thermodynamics : q = $∆$E + W
Since the system is closed and insulated, q = 0
Paddle work is done on system. ∴ W ≠ 0.
Temperature and hence internal energy of the system increases. ∴ $∆$E ≠ 0.
Equilibrium
$A_{2(g)}+B_{2(g)} ⇄ 2AB_{(g)}$
At equilibrium, the concentration of
$A_2=3.0×10^{-3}M$, of $B_2=4.2× 10^{-3}M$, of AB = 2.8 $×10^{-3}$M
If the reaction takes place in a sealed vessel at 527°C, then the value of $K_C$ will be
(1) 2.0
(2) 1.9
(3) 0.62
(4) 4.5
(3) 0.62
$A_{2(g)}+B_{2(g)} ⇄ 2AB_{(g)}$
$K_C=\frac{[AB^2]}{[A_2][B_2]}$
= $\frac{(2.8× 10^{-3})^2}{(3.0×10^{-3})(4.2× 10^{-3})}$ = $\frac{2.8×2.8}{3.0×4.2}$ = 0.62
Redox Reactions
(1) Evaporation of $H_2O$
(2) Both oxidation and reduction
(3) $H_2SO_4$ with NaOH
(4) In atmosphere $O_3$ from $O_2$ by lightning
(2) Both oxidation and reduction
Redox reactions are those chemical reactions which involve both oxidation and reduction simultaneously.
Hydrogen
(1) synthetic resins method
(2) Calgon’s method
(3) Clark’s method
(4) ion-exchange method.
(3) Clark’s method
Clark’s process is used to remove temporary hardness of water. In this method quick lime is added. The bicarbonates present in temporary hard water react with lime water to form insoluble calcium and magnesium carbonates which can be easily filtered off.
$Cao+H_2O → Ca(OH)_2$
$Ca(OH)_2 + Ca(HCO_3)_2 → 2CaCO_3 + 2H_2O$
$Mg(HCO_3)_2+2Ca(OH)_2 → 2CaCO_3 + Mg(OH)_2 + 2H_2O$
The S-Block Elements
(1) K
(2) Rb
(3) Li
(4) Na
(3) Li
The hydration enthalpy of alkali metal ions decreases with increase in ionic sizes i.e.,
$Li^+> Na^+>K^+>Rb^+>Cs^+$
Hence, lithium having maximum degree of hydration will be least mobile.
The order of ionic mobility is
$[Li_{(aq)}]^+> [Na_{(aq)}]^+>[K_{(aq)}]^+>[Rb_{(aq)}]^+$
The p-Block Elements
(1) $B < Al < In < Ga < Tl$
(2) $B < Al < Ga < In < Tl$
(3) $B < Ga < Al < Tl < In$
(4) $B < Ga < Al < In < Tl$
(4) $B < Ga < Al < In < Tl$
Organic Chemistry - Some Basic Principles & Techniques
(1) homocyclic but not aromatic
(2) aromatic but not homocyclic
(3) homocyclic and aromatic
(4) heterocyclic and aromatic.
(1) homocyclic but not aromatic
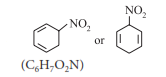
Hence, it is homocyclic (as the ring system is made of one type of atoms, i.e., carbon) but not aromatic.
Hydrocarbons
(1) oxidation
(2) cracking
(3) distillation under reduced pressure
(4) hydrolysis
(2) cracking
The process of cracking converts higher alkanes into smaller alkanes and alkenes. This process can be used for production of natural gas.
Environmental Chemistry
(1) Ozone
(2) Acrolein
(3) Peroxyacetyl nitrate
(4) Chlorofluorocarbons
(4) Chlorofluorocarbons
Solid State
(1) allotropic crystals
(2) liquid crystals
(3) isomeric crystals
(4) isomorphous crystals
(2) liquid crystals
Liquid crystals on heating first become turbid and then on further heating turbidity completely disappears.
Solutions
(1) 10 g
(2) 2 g
(3) 1 g
(4) 20 g
(3) 1 g
The strength of the solution is 0.1 N.
$\frac{w}{E}=\frac{V×N}{1000}$
Equivalent weight = 100
w = $\frac{100× 0.1× 100}{1000}$ = 1 g
Electrochemistry
(i) EMF of cell = (Oxidation potential of anode) – (Reduction potential of cathode)
(ii) EMF of cell = (Oxidation potential of anode) + (Reduction potential of cathode)
(iii) EMF of cell = (Reductional potential of anode) + (Reduction potential of cathode)
(iv) EMF of cell = (Oxidation potential of anode) – (Oxidation potential of cathode)
Which of the above relations are correct?
(1) (iii) and (i)
(2) (i) and (ii)
(3) (iii) and (iv)
(4) (ii) and (iv)
(4) (ii) and (iv)
EMF of a cell = Reduction potential of cathode – Reduction potential of anode
= Reduction potential of cathode + Oxidation potential of anode
= Oxidation potential of anode – Oxidation potential of cathode.
Chemical Kinetics
(1) 12
(2) 16
(3) 32
(4) 10
(3) 32
Rate$_1$ = $k[A]^2 [B]^3$
Rate$_2$ = $k[2A]^2[2B]^3$
Rate$_2$ = $32k[A]^2[B]^3$
∴ Rate$_2$ = 32(Rate$_1$)
Surface Chemistry
(1) $∆$G and $∆$H are negative but $∆$S is positive.
(2) $∆$G and $∆$S are negative but $∆$H is positive.
(3) $∆$G is negative but $∆$H and $∆$S are positive.
(4) $∆$G, $∆$H and $∆$S all are negative.
(4) $∆$G, $∆$H and $∆$S all are negative.
As the molecules of the adsorbate are held on the surface of the solid adsorbent, entropy decreases i.e., $∆$S = –ve.
As $∆$G = $∆$H – T$∆$S
For the adsorption to occur, $∆$G = –ve and it is possible only if $∆$H = –ve.
General Principles & Processes in Isolation of Elements
(1) $CO_2 $
(2) $SO_3$
(3) $H_2S $
(4) $SO_2 $
(4) $SO_2 $
The p - Block Elements
(1) its atom has a stable electronic configuration
(2) it has low atomic radius
(3) its electronegativity is fairly high
(4) dissociation energy of its molecule is fairly high
(4) dissociation energy of its molecule is fairly high
$N_2$ molecule contains triple bond between N atoms having very high dissociation energy (946 kJ mol$^{–1}$) due to which it is relatively inactive.
The D & F - Block Elements
(1) $Fe^{2+}, Ni^{2+} $
(2) $Zr^{4+}, Ti^{4+}$
(3) $Zr^{4+}, Hf^{4+} $
(4) $Zn^{2+}, Hf^{4+}$
(3) $Zr^{4+}, Hf^{4+} $
$Hf^{4+}$ and $Zr^{4+}$ belong to group IVB. But, $Hf^{4+}$ has same size as $Zr^{4+}$ due to the addition of 14 lanthanide elements before it in which electrons are added into the f-subshell which poorly shield the outer electrons and contraction in size occurs.
Coordination Compounds
(1) $CH_3NH_2$
(2) $CH_3CN$
(3) Br
(4) $C_2O_4^{2-}$
(4) $C_2O_4^{2-}$
When a ligand has two groups that are capable of bonding to the central atom, it is said to be bidentate. Thus, the only ligand,which is expected to be bidentate is $C_2O_4^{2-}$
Haloalkanes & Haloarenes
(1) PVC
(2) Allyl chloride
(3) Propyl chloride
(4) 1, 2-Dichloroethane
(2) Allyl chloride
$CH_3CH=CH_2+Cl_2-HCl → ClCH_2CH=CH_2$
At 400°C temperature, substitution occurs instead of addition.
Alcohols, Phenols & Ethers
(1) a carboxylic acid
(2) an aldehyde
(3) a ketone
(4) an alkene
(3) a ketone

Aldehydes, Ketones & Carboxylic Aids
(1) Etard reaction
(2) Riemer–Tiemann reaction
(3) Wurtz reaction
(4) Cannizzaro’s reaction
(1) Etard reaction
The oxidation of toluene ($C_6H_5CH_3$) with chromyl chloride ($CrO_2Cl_2$) in $CCl_4$ or $CS_2$ to give benzaldehyde is called Etard reaction. In this reaction, the chromyl chloride first forms a brown complex, which is separated and then decomposed with $H_2O$ to give benzaldehyde ($C_6H_5CHO$).
Amines
(1) Hoffmann hypobromamide reaction
(2) Stephen’s reaction
(3) Gabriel phthalimide synthesis
(4) Carbylamine reaction
(1) Hoffmann hypobromamide reaction

Biomolecules
(1) Glucose
(2) Sucrose
(3) Maltose
(4) Lactose
(2) Sucrose
All monosaccharides whether aldoses or ketoses are reducing sugars. Disaccharides such as sucrose in which the two monosaccharide units are linked through their reducing centres i.e., aldehydic or ketonic groups are non-reducing.
Polymers
(1) alternate cis- and trans-configuration
(2) random cis- and trans-configuration
(3) all cis-configuration
(4) all trans-configuration
(3) all cis-configuration
Natural rubber is cis-polyisoprene.
Chemistry in Everyday Life
(1) antiseptic
(2) antipyretic
(3) antibiotic
(4) analgesic
(1) antiseptic
Dettol which is a well known antiseptic is a mixture of chloroxylenol and a-terpineol in a suitable solvent.
Biomolecules
(1) asymmetric carbons
(2) primary alcoholic group
(3) secondary alcoholic group
(4) enolisation of fructose followed by conversion to aldehyde base
(4) enolisation of fructose followed by conversion to aldehyde base
Under alkaline conditions of the reagent, fructose gets converted into a mixture of glucose and mannose (Lobry de Bruyn van Ekenstein rearrangement) both of which contain the –CHO group and hence, reduce Tollens’ reagent to give silver mirror test.
Units & Measurements
(1) 8%
(2) 2%
(3) 4%
(4) 6%
(4) 6%
$V=\frac{4}{3}πR^3$
Differentiating $\frac{dV}{V}=3\frac{dR}{R}$
Motion in a Straight Line
(1) 9 km/h
(2) 16 km/h
(3) 18 km/h
(4) 48 km/h
(3) 18 km/h
Total distance travelled = s
Total time taken = $\frac{{s}{3}}{10}+\frac{{s}{3}}{20}+\frac{{s}{3}}{60}$
= $\frac{s}{30}+\frac{s}{60}+\frac{s}{180}$
Average speed = $\frac{total distance travelled}{total time taken}$
= $\frac{s}{{s}{18}}$ = 18 km/h
Motion in a Plane - Vectors
(1) Distance
(2) Angular momentum
(3) Heat
(4) Energy
(2) Angular momentum
Since the angular momentum has both magnitude and direction, it is a vector quantity.
Laws of Motion
(1) 0.07 kg/s
(2) 1.4 kg/s
(3) 0.7 kg/s
(4) 10.7 kg/s
Force = $\frac{d(momentum)}{dt}$
= $\frac{d(mv)}{dt}$ = v($\frac{dm}{dt}$)
210 = 300($\frac{dm}{dt}$)
$\frac{dm}{dt}$ = rate of combustion = $\frac{210}{300}$ = 0.7 kg/s
Work, Energy & Power
(1) 0.18 m/$s^2$
(2) 0.2 m/$s^2$
(3) 0.1 m/$s^2$
(4) 0.15 m/$s^2$
(3) 0.1 m/$s^2$
Here, m = 10 g = $10^{–2}$ kg, R = 6.4 cm = 6.4 × $10^{–2}$ m, $K_f$ = 8 × $10^{–4}$ J, $K_i$ = 0, $a_t$ = ?
Using work energy theorem,Work done by all the forces = Change in KE
$W_{tangential}+W_{centripetal}=K_f-K_i$
$a_t$ = $\frac{K_f}{4πRm}$ = $\frac{8× 10^{–4}}{4×3.14×6.4×10^{–2}×10^{–2}}
=0.099 $≈ 0.1 $ms^{-2}$
System of Particles & Rotational Motion
(1) 3.0 m
(2) 2.3 m
(3) zero
(4) 0.75 m
(3) zero
As no external force acts on the system, therefore centre of mass will not shift.
Gravitation
(1) move towards each other
(2) move away from each other
(3) will become stationary
(4) keep floating at the same distance between them
(1) move towards each other
Since two astronauts are floating in gravitational free space. The only force acting on the two astronauts is the gravitational pull of their masses, F = $\frac{Gm_1m_2}{r^2}$ which is attractive in nature.Hence they move towards each other.
Mechanical Properties of Solids
(1) ductile and brittle
(2) brittle and ductile
(3) brittle and plastic
(4) plastic and ductile
(2) brittle and ductile
Mechanical Properties of Fluids
(1) The velocity is maximum at the narrowest part of the pipe and pressure is maximum at the widest part of the pipe.
(2) Velocity and pressure both are maximum at the narrowest part of the pipe.
(3) Velocity and pressure both are maximum at the widest part of the pipe.
(4) The velocity is minimum at the narrowest part of the pipe and the pressure is minimum at the widest part of the pipe.
(1) The velocity is maximum at the narrowest part of the pipe and pressure is maximum at the widest part of the pipe.
According to equation of continuity,
Av = constant
Therefore, velocity is maximum at the narrowest part and minimum at the widest part of the pipe.
According to Bernoulli’s theorem for a horizontal pipe,
P + $\frac{1}{2}ρv^2$ = constant
Hence, when a fluid flow across a horizontal pipe of variable area of cross-section its velocity is maximum and pressure is minimum at the narrowest part and vice versa.
Thermal Properties of Matter
(1) 450
(2) 1000
(3) 1800
(4) 225
(3) 1800
According to Stefan-Boltzman law, rate of energy radiated by a black body is given as
$E=σAT^4$ = $σ4πR^2T^4$
Given $E_1$ = 450 W, $T_1$ = 500 K, $R_1$ = 12 cm
$R_2$ = $\frac{R_1}{2}$, $T_2$ = 2$T_1$, $E_2$ = ?
$\frac{E_2}{E_1}$ = $\frac{σ4πR_2^2T_2^4}{σ4πR_1^2T_1^4}$ = $\frac{R_2}{R_1})^2(\frac{T_2}{T_1})^4$
$\frac{E_2}{E_1}$ = $\frac{16}{4}$ = 4
$E_2$ = $E_1$ × 4 = 450 × 4 = 1800 W
Thermodynamics
(1) 6400 J
(2) 5400 J
(3) 7900 J
(4) 8900 J
(3) 7900 J
Heat energy given dQ = dU + dW where dU is the change in internal energy and dW is the work done.
Given dQ = 2 kcal = 2000 × 4.2 J and dW = 500 J
∴ 2000 × 4.2 = dU + 500
⇒ dU = 7900 J
Kinetic Theory
(1) collission on walls will be less
(2) number of collisions per unit time will increase
(3) collisions will be in straight lines
(4) collisions will not change.
(2) number of collisions per unit time will increase
As the temperature increases, the average velocity increases. So, the number collisions per unit time will increase.
Oscillations
(1) periodic but not simple harmonic
(2) simple harmonic but not periodic
(3) period and simple harmonic
(4) neither periodic nor simple harmonic
(1) periodic but not simple harmonic
Waves
(1) energy, momentum and mass
(2) energy
(3) energy and mass
(4) energy and linear momentum
(2) energy
With the propagation of a longitudinal wave, energy alone is propagated.
Motion in a Plane - Projectile
(1) 40 m
(2) 50 m
(3) 60 m
(4) 20 m
(1) 40 m
Here, u = 20 m/s, g = 10 m/$s^2$
For maximum range, angle of projection is q = 45°
∴ $R_{max}$ = $\frac{u^2sin90^o}{g}$ = $\frac{u^2}{g}$
= $\frac{20^2}{10}$ = 40 m
Electric Charges & Field
(1) increased K times
(2) remains unchanged
(3) decreased K times
(4) increases 1/K times
(3) decreased K times
$F_m=\frac{F_0}{K}$
i.e., decreases K times
Electric Potential & Capacitance
(1) 80 V
(2) 800 V
(3) zero
(4) 8 V
(1) 80 V
Potential inside the sphere is the same as that on the surface i.e., 80 V.
Current Electricity
(1) increase in temperature
(2) increase in cross-section area
(3) increase in cross-section and decrease in length
(4) decrease in cross-section area.
(1) increase in temperature
Specific resistance is a property of a material and it increases with the increase of temperature, but not vary with the dimensions (length, cross-section) of the conductor.
Moving Charges & Magnetism
(1) move in a circular path with a decreased speed
(2) move in a circular path with a uniform speed
(3) get deflected in vertically upward direction
(4) move in circular path with an increased speed.
(2) move in a circular path with a uniform speed
When a positively charged particle enters in a region of uniform magnetic field, directed perpendicular to the velocity it experiences a centripetal force which will move it in circular path with a uniform speed.
Magnetism and Matter
(1) the current source
(2) the magnetic field
(3) the lattice structure of the material of the rod
(4) the induced electric field due to the changing magnetic field
(1) the current source
Energy of current source will be converted into gravitational potential energy of the rod.
Electromagnetic Induction
(1) electric heater
(2) induction furnace
(3) magnetic braking in train
(4) electromagnet
(1) electric heater
Electric heater works on the principle of Joule’s heating effect.
Alternating current
(1) ratio of voltage in primary and secondary may be increased
(2) energy losses due to eddy currents may be minimised
(3) the weight of the transformer may be reduced
(4) rusting of the core may be prevented.
(2) energy losses due to eddy currents may be minimised
The core of a transformer is laminated to minimise the energy losses due to eddy currents.
Electromagnetic Waves
(1) The radiation emitted is in the infrared region.
(2) The radiation is emitted only during the day.
(3) The radiation is emitted during the summers and absorbed during the winters.
(4) The radiation emitted lies in the ultraviolet region and hence is not visible.
(1) The radiation emitted is in the infrared region.
Every body at all time, at all temperatures emit radiation (except at T = 0), which fall in the infrared region.
Ray Optic & Optic Instruments
(1) scattering
(2) dispersion
(3) reflection
(4) refraction
(1) scattering
According to Rayleigh, the amount of scattering is inversely proportional to the fourth power of the wavelength.
Wave Optics
(1) Refraction
(2) Reflection
(3) Diffraction
(4) Origin of spectra
(4) Origin of spectra
Huygen’s construction of wavefront does not apply to origin of spectra which is explained by quantum theory.
Dual Nature of Matter & Radiation
(1) The photocurrent increases with intensity of light.
(2) The stopping potential increases with increase of incident light.
(3) The current in photocell increases with increasing frequency.
(4) The photocurrent is proportional to the applied voltage.
(1) The photocurrent increases with intensity of light.
Since the emission of photoelectrons is directly proportional to the intensity of the incident light, therefore photocurrent increases with the intensity of light.
Atoms
(1) 1 : 1
(2) 1 : –1
(3) 2 : –1
(4) 1 : –2
(2) 1 : –1
In a Bohr orbit of the hydrogen atom, Kinetic energy = – (Total energy)So, Kinetic energy : Total energy = 1 : –1
Nuclei
(1) increases with mass number at low mass numbers
(2) decreases with mass number at low mass numbers
(3) increases with mass number at high mass numbers
(4) decreases with mass number at high mass numbers.
(4) decreases with mass number at high mass numbers.
For nuclei having A > 56 binding energy per nucleon gradually decreases.
Semiconductors & Electronic Devices
(1) non metal
(2) metal
(3) insulator
(4) none of these.
3) insulator
Semiconductors are insulators at room temperature.
Nuclei
(1) mass number reduces by 6
(2) mass number reduces by 4 and atomic number remains unchanged
(3) mass number reduces by 2
(4) atomic number reduces by 1
(2) mass number reduces by 4 and atomic number remains unchanged
1α reduce the mass number by 4 units and atomic number by 2 units, while 1β only increase the atomic number by 1 unit.
Units & Measurements
(1) Relative density
(2) Gravitational constant
(3) Refractive index
(4) Poisson’s ratio
Relative density, refractive index and Poisson’s ratio all the three are ratios, therefore they are dimensionless constants.
Motion in a Straight Line
(1) 10 m
(2) 5 m
(3) 15 m
(4) 20 m
(1) 10 m
As, $v^2 = u^2 – 2gh$
After reaching maximum height velocity becomes zero.
0 = $(10)^2-2×10×\frac{h}{2}$
∴ h = $\frac{200}{20}$ = 10m
Motion in a Plane
(1) 1 : 1
(2) $r_A$ : $r_B$
(3) $v_A$ : $v_B$
(4) $r_B$ : $r_A$
(1) 1 : 1
Time period, T = $\frac{2π}{ω}$
As $T_A$ = $T_B$
S0, $\frac{2π}{ω_A}$ = $\frac{2π}{ω_B}$
or $ω_A:ω_B$ = 1:1
Laws of Motion
(1) 15 kg
(2) 20 kg
(3) 10 kg
(4) 5 kg
(3) 10 kg
Force (F) = 10 N and acceleration (a) = 1 m/$s^2$.
Mass (m) = $\frac{F}{a}$ = $\frac{10}{1}$ = 10 kg
Work. Energy & Power
(1) 324 J
(2) 486 J
(3) 256 J
(4) 524 J
(2) 486 J
According to law of conservation of linear momentum,
30 × 0 = 18 × 6 + 12 × v
⇒ –108 = 12v ⇒ v = – 9 m/s.
Negative sign indicates that both fragments move in opposite directions.
K.E. of 12 kg = $\frac{1} {2}mv^2$ = $\frac{1}{2}×12×81$ = 486 J
System of Particles & Rotational Motion
(1) position of the particles
(2) relative distances between the particles
(3) masses of the particles
(4) forces acting on the particle
(4) forces acting on the particle
The resultant of all forces, on any system of particles, is zero. Therefore their centre of mass does not depend upon the forces acting on the particles.
Gravitation
(1) 11.2 km/s
(2) 22.4 km/s
(3) 5.6 km/s
(4) 44.8 km/s
(2) 22.4 km/s
$v_e$ = $√(2gR_e)$ = $√(\frac{2GM}{R_e})$
∵ $R_p$ = $\frac{1}{4}R_e$
$v_p$ = 2$v_e$ = 2 × 11.2 = 22.4 km/s
Mechanical Properties of Solids
(1) length = 200 cm, diameter = 2 mm
(2) length = 300 cm, diameter = 3 mm
(3) length = 50 cm, diameter = 0.5 mm
(4) length = 100 cm, diameter = 1 mm
(3) length = 50 cm, diameter = 0.5 mm
Young’s modulus,
Y = $\frac{FL}{A∆L}$ = $\frac{4FL}{πD^2∆L}$
or $∆L$ = $\frac{4FL}{πD^2Y}$
where F is the force applied, L is the length, D is the diameter and DL is the extension of the wire respectively. As each wire is made up of same material therefore their Young’s modulus is same for each wire.
For all the four wires, Y, F (= tension) are the same.
∴ $∆L ∝\frac{L}{D^2$}$
In (1) $\frac{L}{D^2$}$ = $\frac{200 cm}{(0.2cm)^2}$ = $5×10^3cm^{-1}$
In (2) $\frac{L}{D^2$}$ = $\frac{300 cm}{(0.3cm)^2}$ = $3.3×10^3cm^{-1}$
In (3) $\frac{L}{D^2$}$ = $\frac{50 cm}{(0.05cm)^2}$ = $20×10^3cm^{-1}$
In (4) $\frac{L}{D^2$}$ = $\frac{100 cm}{(0.1cm)^2}$ = $10×10^3cm^{-1}$
Mechanical Properties of Fluids
(1) water rises upto a point a little below the top and stays there
(2) water does not rise at all
(3) water rises upto the tip of capillary tube and then starts overflowing like a fountain
(4) water rises upto the top of capillary tube and stays there without overflowing.
(4) water rises upto the top of capillary tube and stays there without overflowing.
Water will not overflow but will change its radius of curvature.
Thermal properties of Matter
(1) black lamp
(2) cavity maintained at constant temperature
(3) platinum black
(4) a lump of charcoal heated to high temperature
(2) cavity maintained at constant temperature
An ideal black body is one which absorbs all the incident radiation without reflecting or transmitting any part of it.
Black lamp absorbs approximately 96% of incident radiation.An ideal black body can be realized in practice by a small hole in the wall of a hollow body (as shown in figure) which is at uniform temperature. Any radiation entering the hollow body through the holes suffers a number of reflections and ultimately gets completely absorbed. This can be facilitated by coating the interior surface with black so that about 96% of the radiation is absorbed at each reflection. The portion of the interior surface opposite to the hole is made conical to avoid the escape of the reflected ray after one reflection.
Thermodynamics
(1) adiabatic
(2) equal in all cases
(3) isothermal
(4) isobaric
(4) isobaric
According to first law of thermodynamics
∆Q = ∆U + ∆W
where,
∆Q = Heat supplied to the system
∆U = Increase in internal energy of the system
∆W = Work done by the system
For an adiabatic process ∆Q = 0 ∴ ∆U = – ∆W
For an isothermal process
∆U = 0 ∴ ∆Q = ∆W
Hence, option (a) is true.
Kinetic Theory of Gases
(1) 9/7
(2) 7/5
(3) 8/7
(4) 5/7
(2) $\frac{7}{5}$
Molar specific heat at constant pressure
$C_p$ = (7/2)R
∴ $C_v$ = $C_p$ - R = (7/2)R - R = (5/2)R
∴ $\frac{C_p}{C_v}$ = $\frac{(7/2)R}{(5/2)R}$ = $\frac{7}{5}$
Oscillation
(1) moving down with uniform acceleration
(2) moving around the earth in geostationary orbit
(3) moving up with uniform velocity
(4) moving up with uniform acceleration
(4) moving up with uniform acceleration
Period of oscillation, T = $2π√(\frac{l}{g})$. Therefore T will decrease when acceleration (g) increases. And g will increase when the rocket moves up with a uniform acceleration.
Waves
(1) 20 m/s
(2) 40 m/s
(3) 5 m/s
(4) 10 m/s
(1) 20 m/s
Frequency (ν) = 100 Hz and distance from fixed end = 10 cm = 0.1 m. When a stationary wave is produced, the fixed end behaves as a node. Thus wavelength (λ) = 2 × 0.1 = 0.2 m. Therefore velocity v = νλ = 100 × 0.2 = 20 m/s.
System of Particles & Rotational Motion
(1) total kinetic energy is conserved
(2) the angular momentum of the sphere about the point of contact with the plane is conserved
(3) only the rotational kinetic energy about the centre of mass is conserved
(4) angular momentum about the centre of mass is conserved.
(2) the angular momentum of the sphere about the point of contact with the plane is conserved
Angular momentum about the point of contact with the surface includes the angular momentum about the centre. Because of friction, linear momentum will not be conserved.
Electric Charges and Fields
(1) increase four times
(2) be reduced to half
(3) remains the same
(4) be doubled
(3) remains the same
According to Gauss's law
$φ_E=\frac{Q_{enclosed}}{ε_0}$
If the radius of the Gaussian surface is doubled, the outward electric flux will remain the same. This is because electric flux depends only on the charge enclosed by the surface.
Electric Potential & Capacitance
(1) becomes zero
(2) increases
(3) decreases
(4) remains same
(2) increases
In bringing an electron towards another electron, work has to be done (since same charges repel each other). The work done stored as electrostatic potential energy, and hence, electrostatic potential energy of the system increases.
Current Electricity
(1) decreases in copper but increases in silicon
(2) increases in copper but decreases in silicon
(3) increases in both
(4) decreases in both.
(1) decreases in copper but increases in silicon
For metal specific resistance decreases with decrease in temperature whereas for semiconductor specific resistance increases with decrease in temperature.
Moving Charges & Magnetism
(1) low resistance in parallel
(2) high resistance in parallel
(3) low resistance in series
(4) high resistance in series.
(1) low resistance in parallel
To convert a galvanometer into ammeter, one needs to connect a low resistance in parallel so that maximum current passes through the shunt wire and ammeter remains protected.
Magnetism and Matter
(1) ferromagnetic material only
(2) paramagnetic and ferromagnetic materials
(3) diamagnetic material only
(4) paramagnetic material only
(3) diamagnetic material only
Magnetic susceptibility is negative for diamagnetic material only.
Electromagnetic Induction
(1) a metal is kept in varying magnetic field
(2) a metal is kept in steady magnetic field
(3) a circular coil is placed in a magnetic field
(4) current is passed through a circular coil
(1) a metal is kept in varying magnetic field
Eddy currents are produced when a metal is kept in a varying magnetic field.
Alternating Current
(1) 20 V and 2.0 mA
(2) 10 V and 0.5 mA
(3) Zero volt and therefore no current
(4) 20 V and 0.5 mA
(3) Zero volt and therefore no current
Transformer cannot work on dc.
Therefore $V_s$ = 0 and $I_s$ = 0
Electromagnetic Waves
(1) Infra-red rays
(2) Ultra violet rays
(3) X-rays
(4) Radio waves
(1) Infra-red rays
As the electromagnetic radiations from Sun pass through the atmosphere, some of them are absorbed by it while other reach the surface of earth. The range of wavelength which reaches earth lies in infrared region. This part of the radiation from the sun has shorter wavelength and can penetrate through the layer of gases like $CO_2$ and reach earth surface. But the radiation from the earth being of longer wavelength can escape through this layer. As a result the earth surface gets warm. This is known as green house effect.
Ray Optic & Optic Instruments
(1) scattering and refraction
(2) internal reflection and dispersion
(3) reflection only
(4) diffraction and dispersion.
(2) internal reflection and dispersion
The rainbow is an example of the dispersion of sunlight by the water drops in the atmosphere. This is a phenomenon due to a combination of the refraction of sunlight by spherical water droplets and of internal (not total) reflection.
Wave Optics
(1) no interference
(2) interference with brighter bands
(3) interference with dark bands
(4) interference with larger width.
(4) interference with larger width.
In vacuum, l increases very slightly compared to that in air. As $β$ ∝ $λ$, therefore, width of interference fringe increases slightly.
Dual Nature of Matter & Radiation
(1) increasing the potential difference between the anode and filament
(2) increasing the filament current
(3) decreasing the filament current
(4) decreasing the potential difference between the anode and filament.
(1) increasing the potential difference between the anode and filament
Atoms
(1) Electric fire
(2) Neon street sign
(3) Red traffic light
(4) Sun
(2) Neon street sign
Neon street sign is a source of line emission spectrum.
Nuclei
(1) Light nuclei
(2) Heavy nuclei
(3) Element lying in the middle of the periodic table
(4) Middle elements, which are lying on binding energy curve.
(1) Light nuclei
The nuclei of light elements have a lower binding energy than that for the elements of intermediate mass. They are therefore less stable; consequently the fusion of the light elements results in more stable nucleus.
Semiconductors & Electronic Devices
(1) the valence band is partially empty and the conduction band is partially filled
(2) the valence band is completely filled and the conduction band is partially filled
(3) the valence band is completely filled
(4) the conduction band is completely empty.
(1) the valence band is partially empty and the conduction band is partially filled
In semiconductors at room temperature the electrons get enough energy so that they are able to over come the forbidden gap. Thus at room temperature the valence band is partially empty and conduction band is partially filled. Conduction band in semiconductor is completely empty only at 0 K.
T
(1) electrons move from collector to base
(2) holes move from base to emitter
(3) electrons move from base to collector
(4) electrons move from emitter to base.
(3) electrons move from base to collector
In n-p-n transistor, the electrons are majority carriers in emitter, which move from base to collector while using n-p-n transistor as an amplifier.
Nuclei
(1) nuclei break up at high temperature
(2) atoms get ionised at high temperature
(3) kinetic energy is high enough to overcome the coulomb repulsion between nuclei
(4) molecules break up at high temperature
(3) kinetic energy is high enough to overcome the coulomb repulsion between nuclei
Extremely high temperature needed for fusion make kinetic energy large enough to overcome coulomb repulsion between nuclei.
Units & Measurements
(1) km/s
(2) km s
(3) km/s$^2$
(4) km s$^2$
(3) km/s$^2$
Units of b = $\frac{x}{t^2}$ = $\frac{km}{s^2}$
Motion in a straight line
(1) 360 m
(2) 340 m
(3) 320 m
(4) 300 m
(4) 300 m
Here, u = 20 m/s, v = 80 m/s, g = 10 m/s$^2$, h = ?
v$^2$ = u$^2$ + 2gh
⇒ 80$^2$ = 20$^2$ + 2 × 10 × h
Hence, h = 300 m
Motion in a Plane
(1) particle A will reach at ground first with respect to particle B
(2) particle B will reach at ground first with respect to particle A
(3) both particles will reach at ground simultaneously
(4) both particles will reach at ground with same speed
(3) both particles will reach at ground simultaneously
Time required to reach the ground is dependent on the vertical motion of the particle. Vertical motion of both the particles A and B are exactly same. Although particle B has an initial velocity, but that is in horizontal direction and it has no component in vertical (component of a vector at a direction of 90° = 0) direction. Hence they will reach the ground simultaneously.
Laws of Motion
(1) zero
(2) 400 N
(3) 800 N
(4) 1200 N
(4) 1200 N
When the lift is accelerating upwards with acceleration a, then reading on the scale
R = m (g + a) = 80 (10 + 5) N = 1200 N
Work, Energy & Power
(1) 1
(2) 0
(3) ∞
(4) -1
(1) 1
For a perfectly elastic collision, e = 1 and for a perfectly inelastic collision, e = 0.
System of Particles and Rotational Motion
(1) linear momentum
(2) angular momentum
(3) kinetic energy
(4) potential energy
(2) angular momentum
When a child sits on a rotating disc, no external torque is introduced. Hence the angular momentum of the system is conserved. But the moment of inertia of the system will increase and as a result, the angular speed of the disc will decrease to maintain constant angular momentum.
Gravitation
(1) the linear momentum of S remains constant in magnitude
(2) the acceleration of S is always directed towards the centre of the earth
(3) the angular momentum of S about the centre of the earth changes in direction, but its magnitude remains constant
(4) the total mechanical energy of S varies periodically with time
(2) the acceleration of S is always directed towards the centre of the earth
The gravitational force on the satellite S acts towards the centre of the earth, so the acceleration of the satellite S is always directed towards the centre of the earth.
Mechanical Properties of Solids
(1) ∆l versus $\frac{1}{l}$
(2) ∆l versus l$^2$
(3) ∆l versus $\frac{1}{l^2}$
(4) ∆l versus l
(2) ∆l versus l$^2$
As V = Al
where A is the area of cross-section of the wire.
Young's Modulus, Y = $\frac{F\A}{∆l/l}$ = $\frac{Fl}{A∆l}$
∆l = $\frac{Fl}{YA}$ = $\frac{Fl^2}{YV}$
$∆l∝l^2$
Hence, the graph between ∆l and $l^2$ is a straight line.
Mechanical Properties of Fluids
(1) $\frac{VR^2}{n^3r^2}$
(2) $\frac{V^2R}{nr}$
(3) $\frac{VR^2}{n^2r^2}$
(4) $\frac{VR^2}{nr^2}$
(4) $\frac{VR^2}{nr^2}$
Let the speed of the ejection of the liquid through the holes be v. Then according to the equation of continuity, $πR^2V$ = $nπr^2v$ or v = $\frac{πR^2V}{nπr^2}$ = $\frac{VR^2}{nr^2}$
Thermal Properties of Matter
(1) (500)$^3$
(2) (500)$^4$
(3) 500
(4) (500)$^2$
(2) (500)$^4$
Temperature of black body T = 500 K Therefore total energy emitted by the black body E ∝ $T^4$ ∝ $(500)^4$
Thermodynamics
(1) it is impossible
(2) it is possible but less probable
(3) it is quite probable
(4) data are incomplete
(1) it is impossible
Efficiency is maximum in Carnot engine which is an ideal engine.
η = $\frac{400-300}{400}×100%$ = 25%
∴ efficiency 26% is impossible for his heat engine.
Kinetic Theory of Gases
(1) diatomic
(2) mixture of diatomic and polyatomic molecules
(3) monoatomic
(4) polyatomic
(3) monoatomic
Since $\frac{R}{C_V}$ = 0.67
$\frac{C_P-C_V}{C_V}$ = 0.67
γ = 1.67 = 5/3, Hence gas is monoatomic.
Oscillations
(1) damping force is small
(2) restoring force is small
(3) applied periodic force is small
(4) quality factor is small
(1) damping force is small
Smaller damping gives a taller and narrower resonance peak.
Waves
(1) 4 s
(2) 5 s
(3) 12 s
(4) 3 s
(3) 12 s
Beats are produced. Frequency of beats will be $\frac{1}{3}-\frac{1}{4}$ = $\frac{1}{12}$ per second
Hence time period = 12 s
Oscillations
(1) $ω_1$ ≠ $ω_0$ and $ω_2$ = $ω_0$
(2) $ω_1$ = $ω_0$ and $ω_2$ = $ω_0$
(3) $ω_1$ = $ω_0$ and $ω_2$ ≠ $ω_0$
(4) $ω_1$ ≠ $ω_0$ and $ω_2$ ≠ $ω_0$
(2) $ω_1$ = $ω_0$ and $ω_2$ = $ω_0$
The amplitude and velocity resonance occurs at the same frequency.
At resonance, i.e., $ω_1$ = $ω_0$ and $ω_2$ = $ω_0$, the amplitude and energy of the particle would be maximum.
Electric Charges & Fields
(1) zero
(2) E
(3) E/2
(4) E/3
(1) zero
Electric field inside the charged spherical shell is zero as there is no charge inside it.
Electrostatic Potential & Capacitance
(1) zero
(2) 0.5 N/C
(3) 1 N/C
(4) 5 N/C
(1) zero
Electric Field in a region, E = $\frac{-dV}{dr}$
But here electric potential is constant. Therefore electric field will be zero.
Current Electricity
(1) fuse
(2) conductor
(3) inductor
(4) switch
(1) fuse
Fuse is an electrical safety device that operates to provide overcurrent protection to an electrical circuit.
Moving Charges & Magnetism
(1) in a circular orbit
(2) along a parabolic path
(3) along a straight line
(4) in an elliptical orbit.
(1) in a circular orbit
Electron travelling in a magnetic field perpendicular to its velocity follows a circular path.
Magnetism and Matter
(i) A is feebly repelled
(ii) B is feebly attracted
(iii) C is strongly attracted
(iv) D remains unaffected
Which one of the following is true?
(1) B is of a paramagnetic material
(2) C is of a diamagnetic material
(3) D is of a ferromagnetic material
(4) A is of a non-magnetic material
(1) B is of a paramagnetic material
Diamagnetic will be feebly repelled.
Paramagnetic will be feebly attracted.
Ferromagnetic will be strongly attracted.
Therefore, A is of diamagnetic material.
B is of paramagnetic material.
C is of ferromagnetic material.
D is of non-magnetic material.
Electromagnetic Induction
(1) remain unchanged
(2) be halved
(3) be doubled
(4) become four times
(4) become four times
Self inductance of a solenoid = $μ_0n^2Al$
where n is the number of turns per length.
So self induction ∝ $n^2$
So inductance becomes 4 times when n is doubled.
Alternating Current
(1) only multiple strands for A.C., either for D.C.
(2) only multiple strands for A.C., only single strand for D.C.
(3) only single strand for D.C., either for A.C.
(4) only single strand for A.C., either for D.C.
(1) only multiple strands for A.C., either for D.C.
The major portion of the A.C. flows on the surface of the wire. So where a thick wire is required, a number of thin wires are joined together to give an equivalent effect of a thick wire. Therefore multiple strands are suitable for transporting A.C. Similarly multiple strands can also be used for D.C.
Electromagnetic Waves
(1) it stops ultraviolet rays
(2) ozone layer reduces green house effect
(3) ozone layer reflects radio waves
(4) ozone layer controls $\frac{O_2}{H_2}$ ratio in atmosphere.
(1) it stops ultraviolet rays
The ozone layer absorbs the harmful ultraviolet rays coming from sun.
Ray Optic & Optic Instruments
(1) Rainbow is a combined effect of dispersion, refraction and reflection of sunlight.
(2) When the light rays undergo two internal reflections in a water drop, a secondary rainbow is formed.
(3) The order of colours is reversed in the secondary rainbow.
(4) An observer can see a rainbow when his front is towards the sun.
(4) An observer can see a rainbow when his front is towards the sun.
An observer can see a rainbow only when his back is towards the sun.
Wave Optics
(1) Interference
(2) Diffraction
(3) Coherence
(4) Polarisation
(4) Polarisation
Sound waves can not be polarised as they are longitudinal. Light waves can be polarised as they are transverse.
Dual Nature of Matter & Radiation
(1) protons
(2) electrons
(3) photons
(4) positrons.
(2) electrons
When a metal is heated, electrons are ejected out of it, which are called thermions.
Atoms
(1) first line of Lyman series
(2) second line of Balmer series
(3) first line of Paschen series
(4) second line of Paschen series.
(2) second line of Balmer series
Jump to second orbit leads to Balmer series. The jump from 4th orbit shall give rise to second line of Balmer series.
Nuclei
(1) They obey the inverse square law of distance.
(2) They obey the inverse third power law of distance.
(3) They are short range forces.
(4) They are equal in strength to electromagnetic forces.
(3) They are short range forces.
Nuclear forces are short range forces.
Semiconductors & Electronic Devices
(1) the cathode
(2) the grid
(3) the glass envelope
(4) the anode
(2) the grid
The amplifying action of a triode is based on the fact that a small change in grid voltage causes a large change in plate current. The AC input signal which is to be amplified is superimposed on the grid potential.
T
(1) it is covalent solid
(2) it has large cohesive energy
(3) high melting point
(4) insoluble in all solvents.
(2) it has large cohesive energy
Diamond is very hard due to large cohesive energy.
Nuclei
(1) Cadmium
(2) Plutonium
(3) Uranium
(4) Heavy water
(4) Heavy water
In nuclear fission, the chain reaction is controlled in such way that only one neutron, produced in each fission, causes further fission. Therefore some moderator is used to slow down the neutrons. Heavy water is used for this purpose.
Disclaimer: Vedanadhi is a voluntary, non-commercial academic inititative solely run by Vishnu Shivam. All resources are offered free of charge unless officially stated otherwise. Any use of the Vedanadhi name or content for commercial, promotional, or consultancy purposes without formal approval is unauthorized and may lead to legal action. Vedanadhi is not affiliated with any external business unless publicly announced. Future changes will be communicated on this site. If you find value in this initiative, consider donating to help improve and sustain the platform as a free resources for all.UPI number: 9500512320 (UPI ID: drvishnushivam@oksbi)
Timetable
| Preparation time (in Days) | Physics | Chemistry | Biology |
|---|---|---|---|
| 02 | Physical world & Units and measurements | Some basic concepts of chemistry | |
| 02 | Motion in a straight line | Living World and Biological Classification | |
| 02 | Motion in a plane | Structure of atom | |
| 02 | Laws of motion | Plant Kingdom | |
| 02 | Work, energy & power | Classification of elements and periodicity in properties | |
| 02 | Motion of system of particles & rigid body | Animal Kingdom | |
| 02 | Gravitation | Chemical bonding and Molecular structure | |
| 02 | Mechanical properties of solids | States of Matter | Plant Morphology and Anatomy |
| 02 | Mechanical properties of fluids | Thermodynamics | |
| 02 | Thermal properties | Structural Organisation in Animals | |
| 02 | Thermodynamics | Equilibrium | |
| 02 | Behaviour of perfect gases & Kinetic theory | Redox reactions | Transport, Mineral Nutrition and Hormones in Plants |
| 02 | Oscillations | Hydrogen | |
| 02 | Waves | S-Block elements | Photosynthesis and Respiration |
| 02 | Class 11 Physics full test | P block elements (Group 13 & 14) | |
| 02 | Organic chemistry – Some basic principles and techniques | Digestion, Circulation and Respiration in Animals | |
| 02 | Electric charges and fields | Hydrocarbons | |
| 02 | Electric potential and capacitance | Environmental chemistry | Locomotion, Neural and Chemical Coordination in Animals |
| 02 | Current Electricity | Chemistry Class 11 full test | |
| 02 | Magnetic Effects of current (Magnetic charges and fields) | Solid State | Reproduction in Organisms and Plants |
| 02 | Magnetism and matter | Solutions | |
| 02 | Electromagnetic induction | Electrochemistry | Reproduction in Animals and Reproductive health |
| 02 | Electromagnetic induction | Surface chemistry | |
| 02 | Electromagnetic waves | Chemical Kinetics | Principles of Inheritance & Variation |
| 02 | Ray Optics | General principles and processes of isolation of elements | |
| 02 | Wave Optics | P block elements (Group 15, 16, 17 & 18) | Molecular Basis of Inheritance |
| 02 | Dual Nature of Matter and Radiation | d and f block elements | |
| 02 | Atoms | Evolution and Ecosystem | |
| 02 | Nuclei | Haloalkanes and Haloarenes | |
| 02 | Semiconductors(Electronic devices) | Aldehydes, Ketones and Carboxylic acids | Biotechnology and its Principles |
| 01 | Organic compounds containing Nitrogen | ||
| 02 | Microbes in Human Welfare(3 lessons) | ||
| 02 | Class 12 Full physics test | Alcohols, Phenols and Ethers | |
| 02 | Biomolecules | Organisms and Population, Biodiversity, Environmental Issues | |
| 01 | Polymers | ||
| 02 | Chemistry in Everyday life | Cell, Cell cycle and Biomolecules | |
| 04 | Physics Full test | ||
| 06 | Chemistry Full Syllabus test | Biology Full Test | |
| 10 | NEET Sample Paper 1 | ||
| 10 | NEET Sample Paper 2 All The Best |
||
| Week No. | Physics | Chemistry | Biology |
|---|---|---|---|
| 001 | |||
| 002 | Living World | ||
| 003 | Physical world, Units & Measurements | ||
| 004 | Some basic concepts of chemistry | Biological Classfication | |
| 005 | |||
| 006 | Motion in a straight line | ||
| 007 | Structure of atom | Plant Kingdom | |
| 008 | |||
| 009 | Motion in a plane | ||
| 010 | Classification of elements and periodicity in properties | Animal Kingdom | |
| 011 | |||
| 012 | Morphology of Flowering Plants | ||
| 013 | Laws of motion | Chemical bonding and Molecular structure | |
| 014 | Anatomy of Flowering Plants | ||
| 015 | States of Matter | ||
| 016 | Structural Organisation in animals | ||
| 017 | Work, Energy & power | cockroach | |
| 018 | Thermodynamics | ||
| 019 | Cell: The unit of life | ||
| 020 | Motion of System of particles & Rigid body | ||
| 021 | Biomolecules | ||
| 022 | Equilibrium | ||
| 023 | Cell cycle & Cell division | ||
| 024 | Gravitation | Redox reactions | |
| 025 | Transport in Higher Plants | ||
| 026 | Mechanical properties of solids | Hydrogen | |
| 027 | Mineral Nutrition in Higher Plants | ||
| 028 | S-block elements | ||
| 029 | Photosynthesis in Higher plants | ||
| 030 | Mechanical properties of fluids | ||
| 031 | Respiration in Higher Plants | ||
| 032 | The P-Block Elements (Group 13 & 14) | ||
| 033 | Plant growth and development | ||
| 034 | Thermal properties | ||
| 035 | Digestion & Absorption in Animals | ||
| 036 | Organic chemistry – Some basic principles and techniques | ||
| 037 | Breathing & Exchange of Gases | ||
| 038 | Thermodynamics | ||
| 039 | Body fluids and circulation | ||
| 040 | Behaviour of perfect gases & kinetic theory | ||
| 041 | Excretory products and their elimination | ||
| 042 | Hydrocarbons | ||
| 043 | Locomotion & Movement in Animals | ||
| 044 | Oscllations | ||
| 045 | Neural control & coordination | ||
| 046 | Environmental chemistry | ||
| 047 | Chemical coordination & Integration | ||
| 048 | Waves | ||
| 049 | |||
| 050 | Class 11 Biology Full test | ||
| 051 | Class 11 Physics full test | ||
| 052 | Class 11 Chemistry Full test | ||
| 053 | Solid State | Reproduction in Organisms | |
| 054 | Sexual reproduction in plants | ||
| 055 | Electric charges and fields | Human reproduction | |
| 056 | Solutions | Reproductive health | |
| 057 | Principles of inheritance and variation | ||
| 058 | Electric potential and capacitance | Molecular basis of inheritance | |
| 059 | Electrochemistry | Evolution | |
| 060 | Human health and diseases | ||
| 061 | Current Electricity | Microbes in human welfare | |
| 062 | Surface chemistry | Strategies for enhancement in food production | |
| 063 | Biotechnology principles and processes | ||
| 064 | Magnetic Effects of current (Magnetic charges and fields) | Biotechnology and its applications | |
| 065 | Chemical Kinetics | Organisms and population | |
| 066 | Ecosystem | ||
| 067 | Magnetism and matter | Biodiversity and conservation | |
| 068 | General principles and processes of isolation of elements | Environmental issues | |
| 069 | |||
| 070 | Electromagnetic induction | Class 12 Full test | |
| 071 | P block elements (Group 15, 16, 17 & 18) | ||
| 072 | Living World and Biological Classification | ||
| 073 | Alternating current | Plant Kingdom | |
| 074 | d and f block elements | Animal Kingdom | |
| 075 | Electromagnetic waves | ||
| 076 | Haloalkanes and Haloarenes | Plant Morphology and Anatomy | |
| 077 | Structural Organisation in Animals | ||
| 078 | Ray Optics | ||
| 079 | Aldehydes, Ketones and Carboxylic acids | Transport, Mineral Nutrition and Hormones in Plants | |
| 080 | Photosynthesis and Respiration | ||
| 081 | Wave Optics | ||
| 082 | Organic compounds containing Nitrogen | Digestion, Circulation and Respiration in Animals | |
| 083 | Locomotion, Neural and Chemical Coordination in Animals | ||
| 084 | Dual Nature of Matter and Radiation | ||
| 085 | Alcohols, Phenols and Ethers | Reproduction in Organisms and Plants | |
| 086 | Reproduction in Animals and Reproductive health | ||
| 087 | Atoms and Nuclei | Biomolecules & Chemistry in Everyday life | |
| 088 | Principes of Inheritance | ||
| 089 | Molecular Basis of Inheritance | ||
| 090 | Semiconductors(Electronic devices) | ||
| 091 | Polymers | Evolution and Ecosystem | |
| 092 | Biotechnology and its Principles | ||
| 093 | Class 12 Full test | ||
| 094 | Microbes in Human Welfare(3 lessons) | ||
| 095 | Chmistry class 12 full test | ||
| 096 | Organisms and Population, Biodiversity, Environmental Issues | ||
| 097 | Physics Full test | ||
| 098 | Cell, Cell cycle and Biomolecules | ||
| 099 | Chemistry full syllabus test | Biology Full Test | |
| 100 | NEET Model Test -1 | ||
| 101 | NEET Model Test -2 | ||
| 102 | NEET Model Test -3 | ||
| 103 | NEET Sample Paper | ||
| 104 | All The Very BEST | ||
| Week No. | Physics | Chemistry | Biology |
|---|---|---|---|
| 001 | Some basic concepts of chemistry | Living World | |
| 002 | Physical world & Units and measurements | Biological Classification | |
| 003 | Motion in a straight line | Structure of atom | Plant Kingdom |
| 004 | Motion in a plane | Animal Kingdom | |
| 005 | Laws of motion | Classification of elements and periodicity in properties | Morphology of Flowering plants |
| 006 | Work, enrgy & power | Anatomy of Flowering Plants | |
| 007 | Motion of system of particles & Rigid body | Chemical bonding and Molecular structure | Structural Organisation in Animals |
| 008 | Gravitation | States of Matter | Cockroach |
| 009 | Mechanical properties of solids | Thermodynamics | Transport in Higher plants |
| 010 | Mechanical properties of fluids | Mineral Nutrition in Higher plants | |
| 011 | Thermal properties | Equilibrium | Photosynthesis in higher plants |
| 012 | Thermodynamics | Redox reactions | Respiration in higher plants |
| 013 | Behaviour of perfect gases & Kinetic theory | Hydrogen | Plant growth & development |
| 014 | Oscillations | S block elements | Digestion & Absorption in Animals |
| 015 | Waves | Breathing and exchange of gases | |
| 016 | The P-Block Elements (Group 13 & 14) | Body fluids & circulation | |
| 017 | Class 11 Physics full test | Excretory products & their elimination | |
| 018 | Organic chemistry – Some basic principles and techniques | Locomotion and movement | |
| 019 | Electric charges & fields | Neural control & coordination | |
| 020 | Electric potential & capacitance | Hydrocarbons | Chemical coordination & integration |
| 021 | Current electricity | Environmental chemistry | |
| 022 | Magnetic Effects of current (Magnetic charges and fields) | Class 11 Full test | |
| 023 | Magnetism and matter | Class 11 chemistry full test | Reproduction in Organisms |
| 024 | Electromagnetic induction | Solid State | Sexual reproduction in plants |
| 025 | Alternating current | Solutions | Human reproduction |
| 026 | Electromagnetic waves | Electrochemistry | Reproductive health |
| 027 | Ray Optics | Surface chemistry | Principles of inheritance and variation |
| 028 | Wave Optics | Chemical Kinetics | Molecular basis of inheritance |
| 029 | Dual Nature of Matter and Radiation | General principles and processes of isolation of elements | Evolution |
| 030 | Atoms and Nuclei | P block elements (Group 15, 16, 17 & 18) | Human health and diseases |
| 031 | Semiconductors(Electronic devices) | d and f block elements | Microbes in human welfare |
| 032 | Strategies for enhancement in food production | ||
| 033 | Class 12 Physics full test | Haloalkanes and Haloarenes | Biotechnology principles and processes |
| 034 | Aldehydes, Ketones and Carboxylic acids | Biotechnology and its applications | |
| 035 | Organic compounds containing Nitrogen | Organisms and population | |
| 036 | Organic compounds containing Nitrogen | Ecosystem | |
| 037 | Biomolecules & Chemistry in Everyday life | Biodiversity and conservation | |
| 038 | Physics Full syllabus test | Polymers | Environmental issues |
| 039 | Chemistry class 12 full test | Class 12 Full test | |
| 040 | Chemistry Full syllabus test | Biology Full test | |
| 041 | Test Series (3 sets) - 1 | ||
| 042 | Test Series (3 sets) - 2 | ||
| 043 | Test Series (3 sets) - 3 | ||
| 044 | NEET Model Test - 1 | ||
| 045 | NEET Model Test - 2 | ||
| 046 | NEET Model Test - 3 | ||
| 047 | NEET Model Test - 4 | ||
| 048 | NEET Model Test - 5 | ||
| 049 | NEET Model Test - 6 | ||
| 050 | NEET Model Test - 7 | ||
| 051 | NEET Sample Paper | ||
| 052 | All The Very Best | ||
| Day No. | Physics | Chemistry | Biology |
|---|---|---|---|
| 01 | Physical world & Units and measurements | Some basic concepts of chemistry | |
| 02 | Motion in a straight line | Living World and Biological Classification | |
| 03 | Motion in a plane | Structure of atom | |
| 04 | Laws of motion | Plant Kingdom | |
| 05 | Work, energy & power | Classification of elements and periodicity in properties | |
| 06 | Motion of system of particles & rigid body | Animal Kingdom | |
| 07 | Gravitation | Chemical bonding and Molecular structure | |
| 08 | Mechanical properties of solids | States of Matter | Plant Morphology and Anatomy |
| 09 | Mechanical properties of fluids | Thermodynamics | |
| 10 | Thermal properties | Structural Organisation in Animals | |
| 11 | Thermodynamics | Equilibrium | |
| 12 | Behaviour of perfect gases & Kinetic theory | Redox reactions | Transport, Mineral Nutrition and Hormones in Plants |
| 13 | Oscillations | Hydrogen | |
| 14 | Waves | S-Block elements | Photosynthesis and Respiration |
| 15 | Class 11 Physics full test | P block elements (Group 13 & 14) | |
| 16 | Organic chemistry – Some basic principles and techniques | Digestion, Circulation and Respiration in Animals | |
| 17 | Electric charges and fields | Hydrocarbons | |
| 18 | Electric potential and capacitance | Environmental chemistry | Locomotion, Neural and Chemical Coordination in Animals |
| 19 | Current Electricity | Chemistry Class 11 full test | |
| 20 | Magnetic Effects of current (Magnetic charges and fields) | Solid State | Reproduction in Organisms and Plants |
| 21 | Magnetism and matter | Solutions | |
| 22 | Electromagnetic induction | Electrochemistry | Reproduction in Animals and Reproductive health |
| 23 | Electromagnetic induction | Surface chemistry | |
| 24 | Electromagnetic waves | Chemical Kinetics | Principles of Inheritance & Variation |
| 25 | Ray Optics | General principles and processes of isolation of elements | |
| 26 | Wave Optics | P block elements (Group 15, 16, 17 & 18) | Molecular Basis of Inheritance |
| 27 | Dual Nature of Matter and Radiation | d and f block elements | |
| 28 | Atoms | Evolution and Ecosystem | |
| 29 | Nuclei | Haloalkanes and Haloarenes | |
| 30 | Semiconductors(Electronic devices) | Aldehydes, Ketones and Carboxylic acids | Biotechnology and its Principles |
| 31 | Organic compounds containing Nitrogen | ||
| 32 | Microbes in Human Welfare(3 lessons) | ||
| 33 | Class 12 Full physics test | Alcohols, Phenols and Ethers | |
| 34 | Biomolecules | Organisms and Population, Biodiversity, Environmental Issues | |
| 35 | Polymers | ||
| 36 | Chemistry in Everyday life | Cell, Cell cycle and Biomolecules | |
| 37 | Physics Full test | ||
| 38 | Chemistry Full Syllabus test | Biology Full Test | |
| 39 | NEET Sample Paper | ||
| 40 | All The Very Best | ||